NEWS
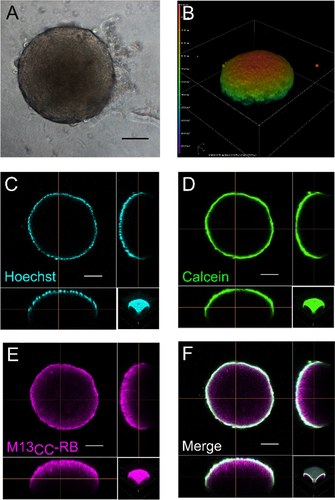
March 2024 - Just published! - Congrats to Molecular Biotechnology Lab and NanoBioInterface Lab authors and collaborators!
Molecular engineering of a spheroid-penetrating phage nanovector for photodynamic treatment of colon cancer cells
Photodynamic therapy (PDT) represents an emerging strategy to treat various malignancies, including colorectal cancer (CC), the third most common cancer type. This work presents an engineered M13 phage retargeted towards CC cells through pentavalent display of a disulfide-constrained peptide nonamer. The M13CC nanovector was conjugated with the photosensitizer Rose Bengal (RB), and the photodynamic anticancer effects of the resulting M13CC-RB bioconjugate were investigated on CC cells. We show that upon irradiation M13CC-RB is able to impair CC cell viability, and that this effect depends on i) photosensitizer concentration and ii) targeting efficiency towards CC cell lines, proving the specificity of the vector compared to unmodified M13 phage. We also demonstrate that M13CC-RB enhances generation and intracellular accumulation of reactive oxygen species (ROS) triggering CC cell death. To further investigate the anticancer potential of M13CC-RB, we performed PDT experiments on 3D CC spheroids, proving, for the first time, the ability of engineered M13 phage conjugates to deeply penetrate multicellular spheroids. Moreover, significant photodynamic effects, including spheroid disruption and cytotoxicity, were readily triggered at picomolar concentrations of the phage vector. Taken together, our results promote engineered M13 phages as promising nanovector platform for targeted photosensitization, paving the way to novel adjuvant approaches to fight CC malignancies.
Turrini E, Ulfo L, Costantini PE, Saporetti R, Di Giosia M, Nigro M, Petrosino A, Pappagallo L, Kaltenbrunner A, Cantelli A, Pellicioni V, Catanzaro E, Fimognari C, Calvaresi M, Danielli A. Molecular engineering of a spheroid-penetrating phage nanovector for photodynamic treatment of colon cancer cells. Cell Mol Life Sci. 2024 Mar 17;81(1):144. doi: 10.1007/s00018-024-05174-7.
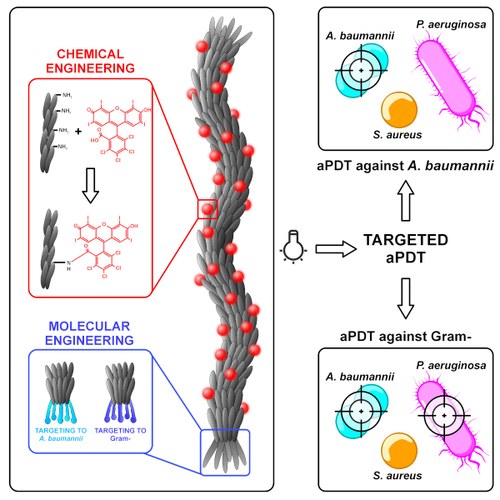
September 2023 - Just published! - Targeted antimicrobial PDT mediated by engineered M13 phages. Congrats to Molecular Biotechnology Lab and NanoBioInterface Lab authors
A modular phage vector platform for targeted photodynamic therapy of Gram-negative bacterial pathogens
Growing antibiotic resistance has encouraged the revival of phage-inspired antimicrobial approaches. On the other hand, photodynamic therapy (PDT) is considered a very promising research domain for the protection against infectious diseases. Yet, very few efforts have been made to combine the advantages of both approaches in a modular, retargetable platform. Here, we foster the M13 bacteriophage as a multifunctional scaffold, enabling the selective photodynamic killing of bacteria. We took advantage of the well-defined molecular biology of M13 to functionalize its capsid with hundreds of photo-activable Rose Bengal sensitizers and contemporarily target this light-triggerable nanobot to specific bacterial species by phage display of peptide targeting moieties fused to the minor coat protein pIII of the phage. Upon light irradiation of the specimen, the targeted killing of diverse Gram(-) pathogens occurred at subnanomolar concentrations of the phage vector. Our findings contribute to the development of antimicrobials based on targeted and triggerable phage-based nanobiotherapeutics.
Petrosino A, Saporetti R, Starinieri F, Sarti E, Ulfo L, Boselli L, Cantelli A, Morini A, Zadran SK, Zuccheri G, Pasquini Z, Di Giosia M, Prodi L, Pompa PP, Costantini PE, Calvaresi M, Danielli A. A modular phage vector platform for targeted photodynamic therapy of Gram-negative bacterial pathogens. iScience. 2023 Sep 27;26(10):108032. doi: 10.1016/j.isci.2023.108032.
FRANCO TATÒ Prize 2023 awarded to Annapaola @SIMGBM Meeting
Annapaola Petrosino, PhD in Cellular and Molecular Biology, was awarded the 2023 "Franco Tatò" Prize for the best doctoral thesis in Microbial Biotechnology at the SIMGBM Meeting, held in Cagliari from 21 to 24 September 2023.
Thesis title: "Nanobiotechnological engineering of M13 phage in an orthogonal platform for biomedical applications".
Congrats to Annapaola!

03-04 May 2022 - Bologna
EIC Pathfinder ECLIPSE Kickoff Meeting
...thrilled to be part of the team !
MBTlab in the News
Press Review
Februrary 2022 - Just published! - PDT review by PhageKiss and NanoBioInterface labs
EGFR-Targeted Photodynamic Therapy
The epidermal growth factor receptor (EGFR) plays a pivotal role in the proliferation and metastatization of cancer cells. Aberrancies in the expression and activation of EGFR are hallmarks of many human malignancies. As such, EGFR-targeted therapies hold significant potential for the cure of cancers. In recent years, photodynamic therapy (PDT) has gained increased interest as a non-invasive cancer treatment. In PDT, a photosensitizer is excited by light to produce reactive oxygen species, resulting in local cytotoxicity. One of the critical aspects of PDT is to selectively transport enough photosensitizers to the tumors environment. Accordingly, an increasing number of strategies have been devised to foster EGFR-targeted PDT. Herein, we review the recent nanobiotechnological advancements that combine the promise of PDT with EGFR-targeted molecular cancer therapy. In this review, we recapitulate the chemistry of the sensitizers and their modes of action in PDT, and summarize the advantages and pitfalls of different targeting moieties, highlighting future perspectives for EGFR-targeted photodynamic treatment of cancer.
Ulfo, L.; Costantini, P.E.; Di Giosia, M.; Danielli, A.; Calvaresi, M. EGFR-Targeted Photodynamic Therapy. Pharmaceutics 2022, 14, 241. https://doi.org/10.3390/pharmaceutics14020241
We're on Nanoscale front cover!
21.01.2022 - The inside front cover of Nanoscale highlights an orthogonal nanoarchitectonics approach to engineer M13 bacteriophages as targeted vectors for efficient photodynamic cancer therapy!
https://pubs.rsc.org/.../articlelanding/2022/nr/d1nr06053h
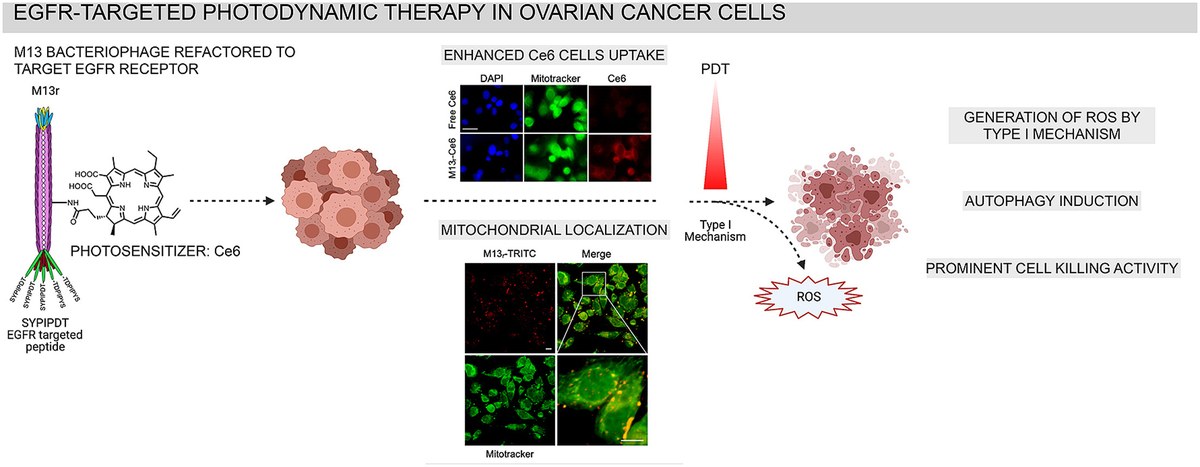
Dec.2021 - Just published! - Congratulations to all PhageKiss and NanoBioInterphase team members for anther joint paper on phage-mediated PDT on ovarian cancer!
Advanced photodynamic therapy with an engineered M13 phage targeting EGFR: Mitochondrial localization and autophagy induction in ovarian cancer cell lines
Photodynamic therapy (PDT) is a potential synergistic approach to chemotherapy for treating ovarian cancer, the most lethal gynecologic malignancy. Here we used M13 bacteriophage as a targeted vector for the efficient photodynamic killing of SKOV3 and COV362 cells. The M13 phage was refactored (M13r) to display an EGFR binding peptide in its tip that is frequently overexpressed in ovarian cancer. The refactored phage was conjugated with chlorin e6 (Ce6), one of the most widely used photosensitizers (M13r-Ce6). The new platform, upon irradiation, generated ROS by type I mechanism and showed activity in killing SKOV3 and COV362 cells even at concentrations in which Ce6 alone was ineffective. A microscopy analysis demonstrated an enhanced cellular uptake of M13r-Ce6 compared to free Ce6 and its mitochondrial localization. Western blot analysis revealed significant downregulation in the expression of EGFR in cells exposed to M13r-Ce6 after PDT. Following PDT treatment, autophagy induction was supported by an increased expression of LC3II, along with a raised autophagic fluorescent signal, as observed by fluorescence microscopy analysis for autophagosome visualization. As a conclusion we have herein proposed a bacteriophage-based receptor targeted photodynamic therapy for EGFR-positive ovarian cancer.

12.11.2021 - Just published! - Congratulations to Luca, Cante and all PhageKiss and NanoBioInterphase team members for first joint paper on phage-mediated PDT!
Orthogonal Nanoarchitectonics of M13 Phage for Receptor Targeted Anticancer Photodynamic Therapy
Photodynamic therapy (PDT) represents a promising therapeutic modality for cancer. Here we used an orthogonal nanoarchitectonics approach (genetic/chemical) to engineer M13 bacteriophages as targeted vectors for efficient photodynamic killing of cancer cells. M13 was genetically refactored to display on the phage tip a peptide (SYPIPDT) able to bind the epidermal growth factor receptor (EGFR). The refactored M13EGFR phages demonstrated EGFR-targeted tropism and were internalized by A431 cancer cells, that overexpress EGFR. Using an orthogonal approach to the genetic display, M13EGFR phages were then chemically modified, conjugating hundreds of Rose Bengal (RB) photosensitizing molecules on the capsid surface, without affecting the selective recognition of the SYPIPDT peptides. Upon internalization, the M13EGFR-RB derivatives generated intracellularly reactive oxygen species, activated by an ultralow intensity white light irradiation. The killing activity of cancer cells is observed at picomolar concentrations of the M13EGFR phage.

September 2021 - Congratulations to Paolo for winning the GENPROBIO 2021 AWARD for best PhD thesis of young SIMGBM members !!
Title of the thesis: "Genomic and functional insights into the interactions between vaginal Lactobacillus strains and pathogens.

18.06.2021 - Just published! - Congratulations to Paolo and all PhageKiss and NanoBioInterphase team members for first joint paper on Photothermal Therapy
Spiky Gold Nanoparticles for the Photothermal Eradication of Colon Cancer Cells
Colorectal cancer (CRC) is a widespread and lethal disease. Relapses of the disease and metastasis are very common in instances of CRC, so adjuvant therapies have a crucial role in its treatment. Systemic toxic effects and the development of resistance during therapy limit the long-term efficacy of existing adjuvant therapeutic approaches. Consequently, the search for alternative strategies is necessary. Photothermal therapy (PTT) represents an innovative treatment for cancer with great potential. Here, we synthesize branched gold nanoparticles (BGNPs) as attractive agents for the photothermal eradication of colon cancer cells. By controlling the NP growth process, large absorption in the first NIR biological window was obtained. The FBS dispersed BGNPs are stable in physiological-like environments and show an extremely efficient light-to-heat conversion capability when irradiated with an 808-nm laser. Sequential cycles of heating and cooling do not affect the BGNP stability. The uptake of BGNPs in colon cancer cells was confirmed using flow cytometry and confocal microscopy, exploiting their intrinsic optical properties. In dark conditions, BGNPs are fully biocompatible and do not compromise cell viability, while an almost complete eradication of colon cancer cells was observed upon incubation with BGNPs and irradiation with an 808-nm laser source. The PTT treatment is characterized by an extremely rapid onset of action that leads to cell membrane rupture by induced hyperthermia, which is the trigger that promotes cancer cell death.
Costantini et al. Nanomaterials (2021) 11:1606
28.04.2021 - Just published! - New article from UniBO team of statisticians, CNR physicist and our lab sheds light on striking (mathematical) properties of the genetic code
A role for circular code properties in translation
Circular codes represent a form of coding allowing detection/correction of frame-shift errors. Building on recent theoretical advances on circular codes, we provide evidence that protein coding sequences exhibit in-frame circular code marks, that are absent in introns. These properties strongly correlate with translation speed, codon influence and protein synthesis levels. The results can lead to defining new universal tools for sequence indicators and sequence optimization for bioinformatics and biotechnological applications, and shed new light on the molecular mechanisms behind the decoding process.
Giannerini et al. Scientific Reports (2021) 11:9218